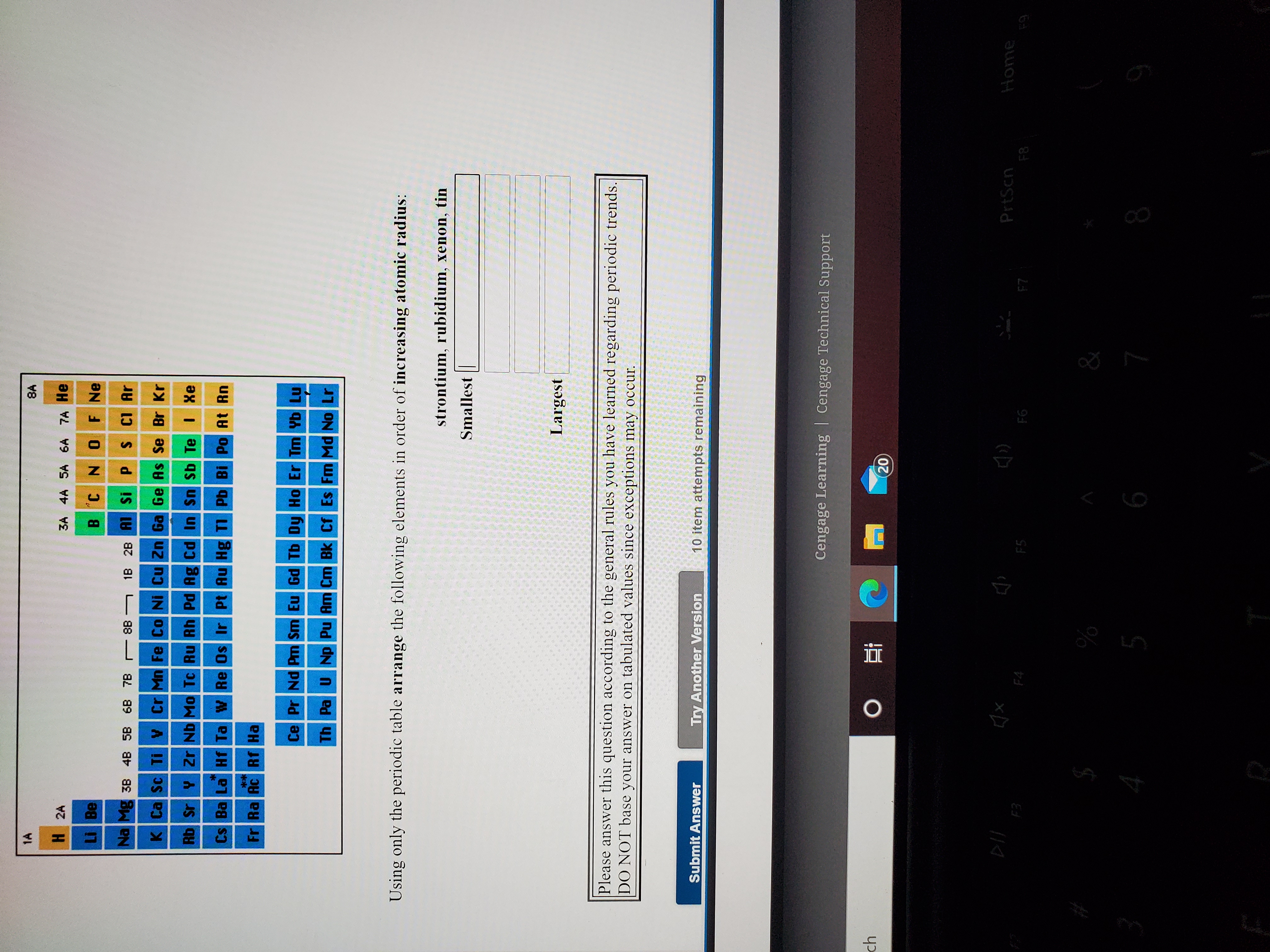
(b) Group VIII like groups I-VII has been divided into two sub-groups A and B. 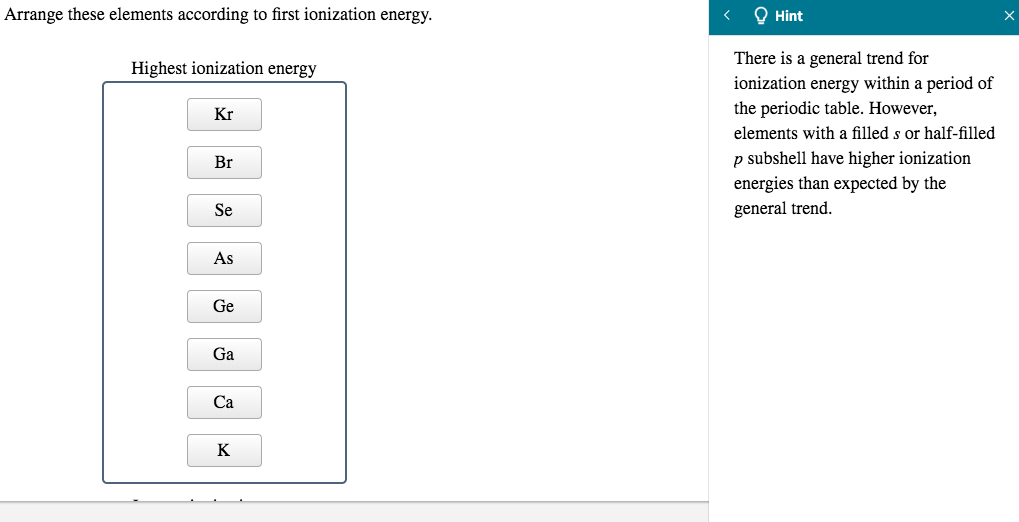
(a) In the mendeleev's periodic table, some places were left vacant for new elements which were not discovered at that time. (v) Which of the following statements is not correct about Mendeleev's periodic table? (iv) The properties of eka-aluminium predicted by Mendeleev were the same as properties of which element that was discovered later? (a) Scandium 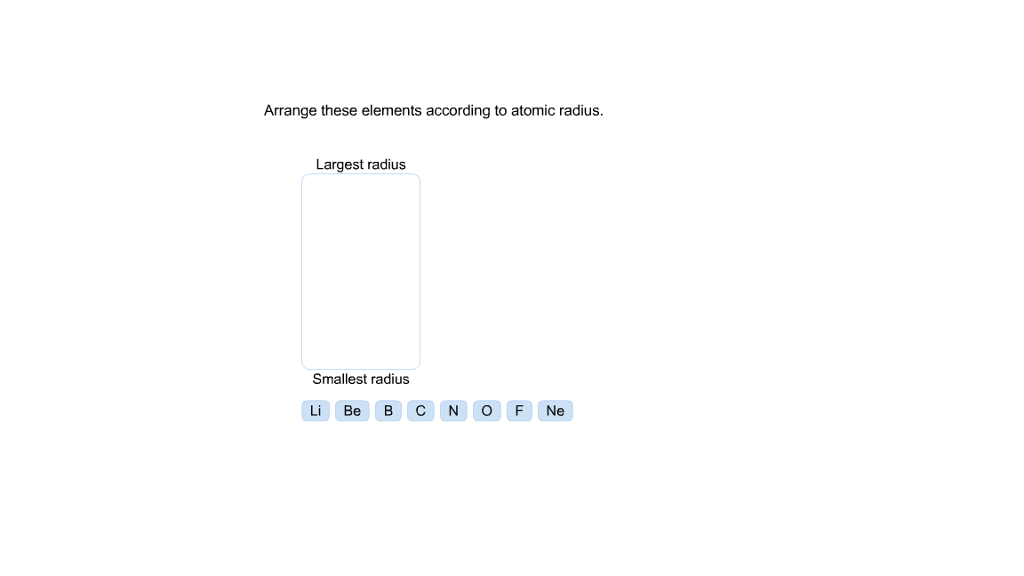
Which of the following elements found a place in the periodic table later? (a) Chlorine (iii) In Mendeleev's periodic table, gaps were left for the elements to be discovered later. The group to which silver belongs in the modern periodic table is (a) first (ii) In Mendeleevs periodic table, silver belongs to IB group. (i) Which of the following metals is not placed in eighth group of Mendeleev's periodic table? (a) Fe (II) The physical and chemical properties of elements are periodic function of their atomic masses. (I) Atomic mass is the fundamental property of elements. The basis of Mendeleev's classification is his periodic law which states that: Mendeleev's periodic table contains vertical columns called 'groups' and horizontal rows called 'periods': Elements with similar properties were placed in same groups. Mendeleev arranged the 63 elements known at that time in order of their ascending atomic masses and prepared a periodic table. (v) On what basis the elements are arranged in Dobereiner's triad? (a) Atomic number (iv) According to the Newlands' law of octaves, the properties of magnesium are similar to those of (a) beryllium How many elements are there in between A and B? (a) 7 (iii) A and B are two elements having similar properties which obey Newlands' law of octaves. (ii) Example of Dobereiner's triad is (a) Li, AI, Ca (i) If CI, Br, I is a Dobereiners triad and the atomic masses of CI and I are 35.5 and 127 respectively, then the atomic mass of Br is (a) 162.5 In 1865, John Newlands arranged all known elements in the order of increasing atomic masses and found that the properties of every eighth element are similar to the properties of the first element. He identified some groups of three elements having similar physical and chemical properties, known as Dobereiners triads. In 1817, Johann Wolfgang Dobereiner tried to arrange the elements with similar properties into groups. After the discovery of large number of elements it became necessary to classify them and arrange them in a regular manner in order of their periodic properties.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
